Adaptive Biotechnologies®

- Partner Level
- Gold
- Company Headquarters
- 1551 Eastlake Ave E, Ste 200, Seattle, WA 98102
- Phone
- (888) 552-8988
- Website
- Visit Site
Overview
About Adaptive Biotechnologies
Adaptive Biotechnologies (“we” or “our”) is a commercial-stage biotechnology company focused on harnessing the inherent biology of the adaptive immune system to transform the diagnosis and treatment of disease. We believe the adaptive immune system is nature’s most finely tuned diagnostic and therapeutic for most diseases, but the inability to decode it has prevented the medical community from fully leveraging its capabilities. Our proprietary immune medicine platform reveals and translates the massive genetics of the adaptive immune system with scale, precision and speed. We apply our platform to partner with biopharmaceutical companies, inform drug development, and develop clinical diagnostics across our two business areas: Minimal Residual Disease (MRD) and Immune Medicine. Our commercial products and clinical pipeline enable the diagnosis, monitoring, and treatment of diseases such as cancer and autoimmune disorders. Our goal is to develop and commercialize immune-driven clinical products tailored to each individual patient.
For more information, visit Adaptive Biotechnologies
About clonoSEQ®
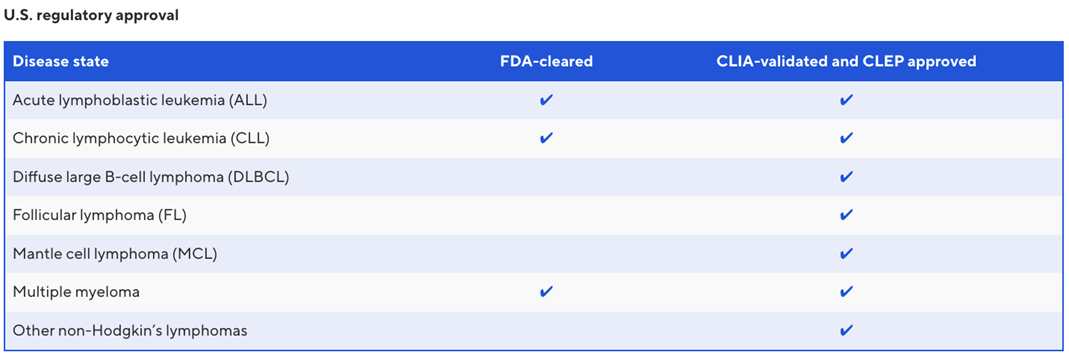
clonoSEQ® is the first and only FDA-cleared in vitro diagnostic (IVD) test for detecting and tracking minimal (or measurable) residual disease (MRD) in patients with multiple myeloma (MM) or B-cell acute lymphoblastic leukemia (B-ALL) using bone marrow, and in patients with chronic lymphocytic leukemia (CLL) using blood or bone marrow. clonoSEQ is also available in diffuse large B-cell lymphoma (DLBCL), mantle cell lymphoma (MCL) and other lymphoid cancers and specimen types as a CLIA-validated laboratory developed test (LDT). clonoSEQ is covered by Medicare for MM, CLL, ALL, DLBCL and MCL.
clonoSEQ identifies and quantifies DNA sequences in malignant cells—detecting one cancer cell in one million healthy cells—to help clinicians and researchers assess and monitor MRD with precision over time. It delivers standardized, sensitive results that inform treatment decisions, predict outcomes, and detect relapses earlier.
For more information, visit clonoSEQ NGS MRD
clonoSEQ MRD test
Comprehensively validated
Widely reimbursed
- 300 million lives covered by Medicare and every major private insurer
- > 90% of clonoSEQ patients pay $0 dollars out of pocket for testing
Broadly adopted by clinicians
- Ordered by more than 6,100 oncologists in 2025
- Available and actively used at 1,800 leading academic and community centers, including all 33 National Comprehensive Cancer Network (NCCN) cancer centers
- More than 82,000 clonoSEQ test results delivered in 2025 to inform treatment decisions
Recommended by practice-defining guidelines
NGS-based MRD testing is incorporated in NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) ALL, CLL, DLBCL, MCL and multiple myeloma
Extensively studied
More than 250 peer-reviewed publications in ALL, CLL, DLBCL, MCL and multiple myeloma
Trusted by biopharma
Used in more than 175 biopharma-sponsored trials, including 15+ studies in which it is used as a primary endpoint to support drug development
Patient Assistance Programs
For clonoSEQ® patients, Adaptive Assist™ is with you every step of the way.
We understand that each patient’s situation is unique,n which is why Adaptive Assist provides support at each point in the insurance and billing process, helping to make each step straightforward and transparent. The Adaptive Assist program helps facilitate access to clonoSEQ testing services for lymphoid cancer patients who could benefit from the clinical insights provided by measurable (or minimal) residual disease (MRD) testing.
We support patients with:
- Upfront, out-of-pocket estimates
- Prior authorization management
- Appeal for maximum benefits and lowest out-of-pocket costs
- Assistance with out-of-pocket costs after coverage for qualifying patients
- 5-minute enrollment with rapid verification
For more details, encourage your patients to call our Patient Support Team at
1 (855) 236-9230 or visit Adaptive-Assist.com